



iSyncWave - AI-Powered EEG brainwave analysis with NIR PBM therapy machine
-
Introducing iSyncWave: A Revolutionary EEG and PBM Solution
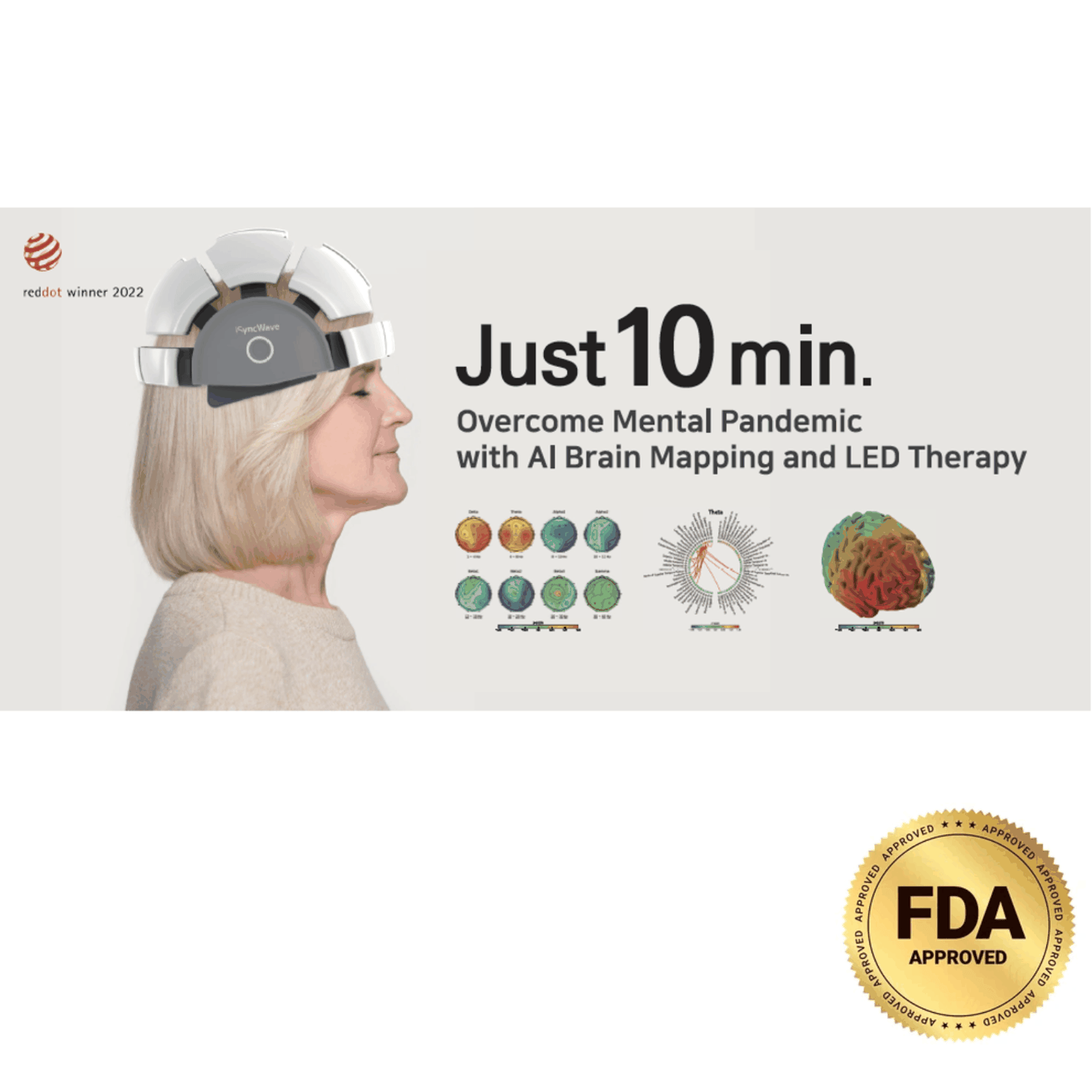
iSyncWave is an advanced, FDA-cleared neurotechnology device that integrates AI-powered EEG brainwave analysis with therapeutic near-infrared (NIR) photobiomodulation (PBM) therapy. Designed for neurologists, psychiatrists, psychologists, mental health practitioners, rehabilitation centers, and holistic health professionals, iSyncWave delivers precise, non-invasive brain assessments and personalized cognitive enhancement solutions. With its wireless, user-friendly design, it empowers healthcare providers to offer advanced brain health diagnostics and therapy, improving patient outcomes with ease and efficiency.
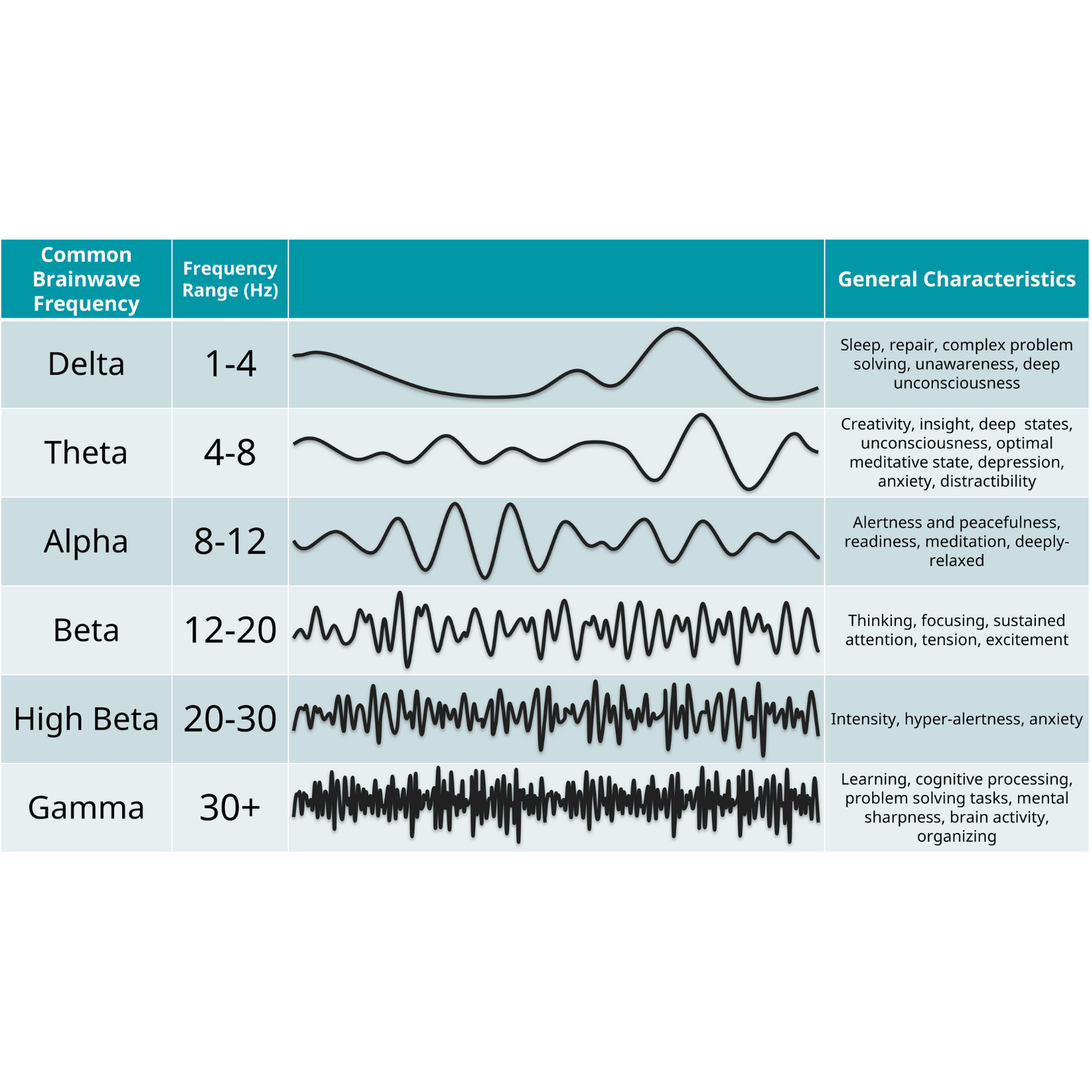
1. Comprehensive Brain Health Monitoring
Equipped with 19 dry EEG channels and a high-precision heart rate variability (HRV) monitor, iSyncWave delivers a full-spectrum neurophysiological assessment to help detect and manage conditions such as:
- Dementia and Alzheimer’s disease
- Depression and anxiety disorders
- PTSD and traumatic brain injuries (TBI)
- Cognitive decline, brain fog, and stress-related conditions
- ADHD and focus-related challenges
2. AI-Powered Precision and Early Detection
Using a vast EEG normative database, iSyncWave’s AI-driven algorithms analyze brainwave patterns to detect early biomarkers of neurological disorders. This empowers healthcare providers to offer preventative insights and create personalized treatment plans for improved patient outcomes.
3. Cutting-Edge Photobiomodulation (PBM) Therapy
iSyncWave integrates Near-Infrared (NIR) LED technology (850 nm) to stimulate brain activity at the cellular level, promoting healing and recovery. PBM therapy has been clinically proven to:
- Reduce oxidative stress and inflammation, improving neural function.
- Enhance mitochondrial function, leading to better energy production in brain cells.
- Promote cellular repair and neuroplasticity, aiding cognitive regeneration and emotional stability.
4. Ease of Use and Accessibility
iSyncWave’s wireless, dry-electrode system eliminates the need for gels or lengthy setup times, making it ideal for a variety of settings, including:
- Clinics and hospitals – Quick and accurate brain assessments.
- Home-use by trained professionals – Convenient treatment sessions.
- Research institutions – Streamlined brain health studies.
5. Scalable Revenue Opportunities for Healthcare Providers
Investing in iSyncWave allows businesses to expand their services and increase revenue streams through:
- Brain health check-ups – Routine scans for proactive care.
- Neurofeedback therapy sessions – Targeted interventions for cognitive improvement.
- Corporate wellness programs – Supporting employee mental well-being.
- Telemedicine consultations – Remote monitoring and cloud-based analysis.
6. Regulatory Compliance and Safety
iSyncWave has achieved FDA Class II clearance (K220056), ISO 13485 certification, and has received the Red Dot Award for innovation, ensuring compliance with the highest standards of safety and effectiveness.
Clinical Studies Supporting iSyncWave’s Effectiveness
Extensive clinical research backs the efficacy of iSyncWave’s PBM and EEG technologies in treating various neurological disorders:
Proven Results for Brain Health Conditions
-
Stroke Recovery
- BMJ Case Reports (2012) showed significant improvements in stroke rehabilitation outcomes through NIR therapy.
-
Traumatic Brain Injury (TBI)
- A study in Photomedicine and Laser Surgery (2016) reported cognitive performance enhancements post-PBM application.
-
Depression and Anxiety
- Research published in Neuropsychiatric Disease and Treatment (2019) indicated significant reductions in depressive symptoms and enhanced emotional stability.
Objective Biomarkers for Mental Health Diagnosis
iSyncWave’s EEG-based biomarkers have been validated for identifying and tracking neurological conditions such as:
- Early-stage dementia and Alzheimer's detection.
- Quantifiable PTSD brainwave abnormalities, enabling more precise treatment strategies.
Supporting PTSD Recovery in First Responders
A Harvard-led study conducted among 380 first responders in Texas demonstrated that iSyncWave’s EEG data significantly correlated with PTSD assessments, showing:
- Marked reductions in alpha2 brain rhythms, which are linked to emotional regulation.
- Improved cognitive function and emotional resilience over time.
Why Invest in iSyncWave?
Psychologists and brain scanner businesses can leverage the iSyncWave to establish themselves as leaders in the field of neurodiagnostics and personalized mental healthcare. Whether used in clinical settings, wellness centers, or research institutions, the device offers unparalleled insights and therapeutic potential with a strong return on investment.
Simple Return On Investmnet (ROI) Breakdown for Patient Consultation Fees:
- Charge $200 - $500 per neuro-diagnostic scan.
- See 50 patients per month, generating $10,000 - $25,000 in monthly revenue.
- Achieve full return on investment (ROI) in 1-3 months, with potential yearly earnings of $120,000 - $300,000.
- By treating 100 patients annually, clinics can help patients save between $500,000 - $2,000,000 through early detection and intervention.
If you're looking to transform your practice and provide cutting-edge brain health solutions, the iSyncWave offers a scientifically backed, AI-powered solution with a robust clinical foundation and a proven track record in mental healthcare innovation.
Net Weight: 4 lbs







